Clik here to view.

An efficient process for the preparation of 1-(2-methoxyphenoxy)-2,3-epoxypropane, a key intermediate for the synthesis of ranolazine is described.
http://pubs.acs.org/doi/suppl/10.1021/op300056k
Image may be NSFW.
Clik here to view.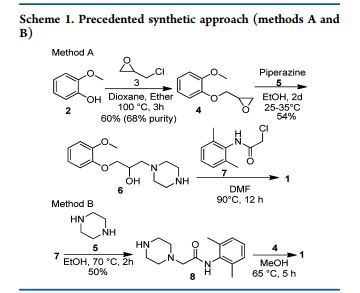
Preparation of 1-(2-Methoxyphenoxy)-2,3-epoxypropane 4.
To a stirring solution of 2-methoxy phenol 2 (10 kg, 80.55 mol) and water (40 L) at about 30 °C was added sodium hydroxide (1.61 kg, 40.25 mol) and water (10 L). After stirring for 30−45 min, epichlorohydrin 3 (22.35 kg, 241.62 mol) was added and stirred for 10−12 h at 25−35 °C. Layers were separated, and water (40 L) was added to the organic layer (bottom layer) containing product. Sodium hydroxide solution (3.22 kg, 80.5 mol) and water (10 L) were added at 27 °C and stirred for 5−6 h at 27 °C.
The bottom product layer was separated and washed with sodium hydroxide solution (3.0 kg 75 mol) and water (30 L). Excess epichlorohydrin (3) was recovered by distillation of the product layer at below 90 °C under vacuum (650−700 mmHg) to give 13.65 kg (94%) of title compound with 98.3% purity by HPLC, 0.2% of 2- methoxy phenol 2, 0.1% of epichlorohydrin 3, 0.1% of chlorohydrin 11, 0.3% of dimer 12 and 0.3% of dihydroxy 13.
1 H NMR (400 MHz, CDCl3, δ) 6.8−7.0 (m, 4H), 4.3 (dd, J = 5.6 Hz, 5.4 Hz, 1H), 3.8 (dd, J = 5.6 Hz, 5.3 Hz, 1H), 3.7 (s, 3H), 3.2−3.4 (m, 1H), 2.8 (dd, J = 5.6 Hz, 5.4 Hz, 1H), 2.7 (dd, J = 5.6 Hz, 5.3 Hz, 1H);
IR (KBr, cm−1 ) 2935 (C−H, aliphatic), 1594 and 1509 (CC, aromatic), 1258 and 1231 (C−O−C, aralkyl ether), 1125 and 1025 (C−O−C, epoxide);
MS (m/z) 181 (M+ + H).
Image may be NSFW.
Clik here to view.
DR REDDYS LABORATORIES
An Efficient Synthesis of 1-(2-Methoxyphenoxy)-2,3-epoxypropane: Key Intermediate of β-Adrenoblockers
////////////////1-(2-Methoxyphenoxy)-2,3-epoxypropane, β-Adrenoblockers, ranolazine